Urticaria, also known as hives or welts, are itchy red bumps that appear on the surface of the skin that fade or blanch when pressure is applied. When it affects the deeper layers of the skin, it can cause swelling or angioedema of the skin, typically affecting the face (eyelid, lips, and tongue), hands, or feet. The underlying mechanism for this lies with a specialized cell called the Mast Cell. Mast cells are found throughout the body, including the surface of our skin. When these mast cells in the skin are stimulated, they release histamine and other allergic mediators that cause hives and swelling. Stimulation of the mast cell can occur from allergic, autoimmune, or unknown triggers. Urticaria can be classified as either acute or chronic.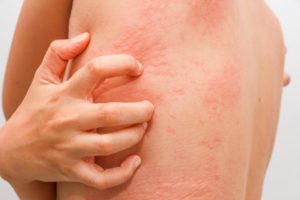
Acute hives
Acute hives are self-limited, usually lasting for a few hours to a few weeks. It can occur in any age group and affects about 25% of people. Common causes include viral infections, foods, medications, or insect stings. It can also be triggered occasionally by heat, exercise or other physical factors. Diagnosis involves a thorough history, physical exam, and testing to help identify the cause of the hives. Treatment may include oral antihistamines to help with itching.
Chronic hives
For a small number of people the hives come back again and again. When this occurs on most days of the week, and persists for more than six weeks, it is called chronic hives. It is more common in adults, especially women, but it can also occur in children as well. About 40% of people with chronic hives have episodes of swelling or angioedema associated with the rash. An identifiable cause of chronic hives is found in only 10% to 20% of patients. The most common cause is an autoimmune reaction generated by a person’s own immune system. These patients may have an underlying autoimmune condition and make autoantibodies against the thyroid gland, the IgE antibody, or the high-affinity IgE receptor. The majority of cases are idiopathic with no known cause. Based on the clinical presentation, diagnostic testing may be necessary to help identify and/or rule-out any underlying disease process that could be associated with the hives. Basic treatment includes non-sedating oral antihistamines, such as Zyrtec (cetirizine), Allegra (fexofenadine), or Claritin (loratadine). Other additional medications include acid suppressors, such as Zantac (ranitidine) which also has antihistamine properties, and leukotriene modifiers, such as Singulair (montelukast). Sedating antihistamines such as Benadryl (diphenhydramine) and Atarax (hydroxyzine) may be needed for breakthrough symptoms. Some patients, however, do not respond to even high doses of these medicines. In these cases, short-term prednisone can be used. But long-term therapy with prednisone is problematic because of the numerous undesirable side effects. Immunosuppressant medications, such as cyclosporine, are effective in treating chronic hives; however, they too have some undesirable side-effects.
Xolair
If high-dose antihistamines are ineffective for sufficient relief, or if frequent oral steroids are needed, Xolair (omalizumab) can be a treatment option. Xolair is an injectable biologic medication originally approved by the FDA in 2003 for the treatment of asthma. In 2014, the FDA approved this medication for the treatment of chronic hives. Like many other biologic medications, Xolair is a monoclonal antibody that works as an “IgE blocker” – it binds to the allergy antibody IgE and forms a complex which is then eliminated from the body. Xolair binds to IgE before IgE attaches to the mast cell. It also reduces the IgE receptors on the surface of mast cells. The end result is less stimulation of mast cells and less release of chemicals that cause hives, itching, and swelling. Therefore, the symptoms and frequency of chronic hives is decreased. In clinical studies, many patients on Xolair were able to reduce the amount of medications they were taking to control their symptoms. This medication is approved for individuals with chronic hives, 12 years and up. It is given as a subcutaneous injection in the office setting on a monthly basis. Approximately 3/4 of patients with chronic urticaria, improve with Xolair, some very dramatically. One study showed that half of patients had complete resolution of their hives.
Common side effects
Based on the clinical studies, the FDA suggests that Xolair is very safe. The most common side effect reported was injection-site reactions. Other adverse reactions reported were colds, sinus infections, headaches and sore throat. Serious adverse reactions occurred in less than 1% of patients. Because it is a biologic medication, there is a risk of anaphylaxis. For this reason, all injections are supervised and given in the office setting.
What’s the next step?
If you or someone you know has experienced hives, the first step would be a thorough history and physical examination. Allergy testing may be done to rule-out allergic causes of the hives. Blood work may be obtained to help identify and rule-out causes for chronic hives. If oral medications are not controlling the hives, Xolair may be a safe long-term option for you to help reduce the frequency and symptoms of chronic hives.
Our physicians at North Texas Allergy & Asthma Center are board-certified allergists with extensive experience in both pediatric and adult allergy, urticaria and Xolair treatment. Call our office for an appointment so we start the evaluation process, answer any questions you may have, and help make you feel better!
SHIKHA MANE, MD
Board Certified Allergist







